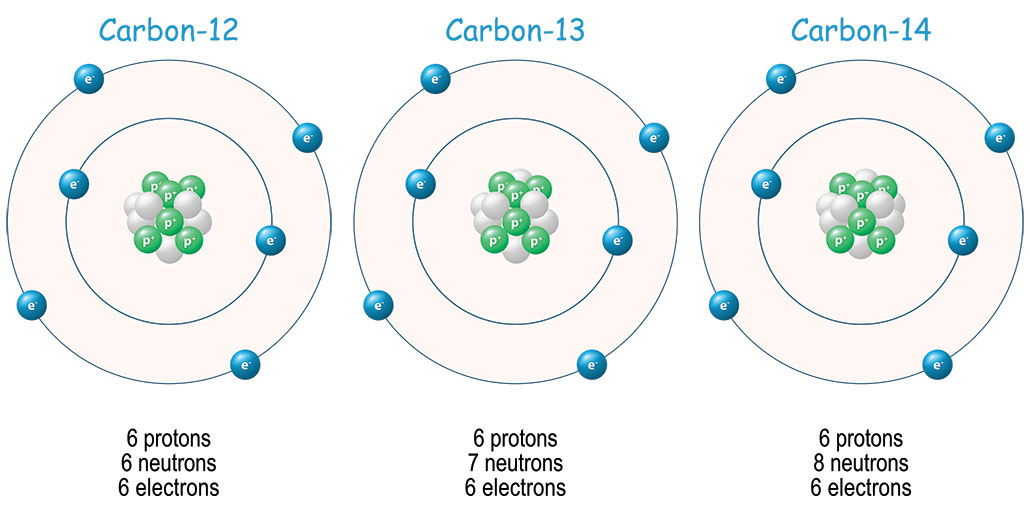
27. Carbon found in nature as a mixture of C 12 C 13. The average atomic mass of carbon is 12.011amu. What is the percentage abundance of carbon 12 in nature

Illustration Chemical Isotopes Carbon All Atoms Stock Vector (Royalty Free) 1717122961 | Shutterstock